- English
- فارسی
A three-layered hollow tubular scaffold as an enhancement of nerve regeneration potential
Nasim Golafshan, Mahshid Kharaziha, Morteza Alehosseini
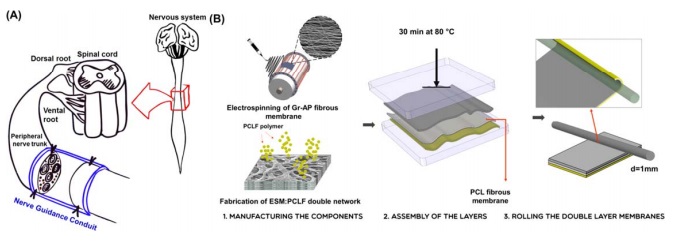
A significant clinical challenge in the surgery of peripheral nervous system injured via accidents and natural disease is development of biomimetic grafts which could potentially promote nerve repair and regeneration. Although various engineered neural tissue scaffolds have been proposed to support the neural cell functions, they have not been able to instantaneously mimic the whole characteristics of endogenous microenvironment. In this study, we proposed a three-layered tubular scaffold which could provide appropriate electrical, mechanical and biological properties for peripheral nerve engineering. While the inter layer was graphene (Gr) embedded alginate-polyvinyl alcohol (AP-Gr) fibrous scaffold with well-defined anisotropy, the outer layer was double network scaffold of polycaprolactone fumarate (PCLF) and eggshell membrane (ESM). These two layers were attached together using a polycaprolactone (PCL) fibrous membrane, a middle layer, via a simple melting process. Results showed that while the electrical conductivity of the three-layered scaffold was similar to that of AP-Gr fibrous layer, the strength of the three-layered scaffold was significantly improved compared to AP-Gr and ESM-PCLF (1.5 and 1.1 times, respectively) attributed to well attachment of the two layers. As a proof-of-concept, PC12 cell attachment, proliferation, and alignment were studied on the developed three-layered scaffold. The majority of the cells (55%) aligned (<20°) along the major axis of fibers features. Furthermore, electrical stimulation revealed positive effect on the alignment of PC12 cells and change in the cell morphology. With the ease of fabrication and mechanical robustness, the three-layered scaffold of AP-Gr and ESM-PCLF might be utilized as a versatile system for the engineering of peripheral nerve tissue.